Laser spectroscopy
For a long time laser spectroscopy has been source of inspiration for EKSPLA scientific laser engineers. Explore some typical applications where our picosecond and nanosecond lasers has been employed.
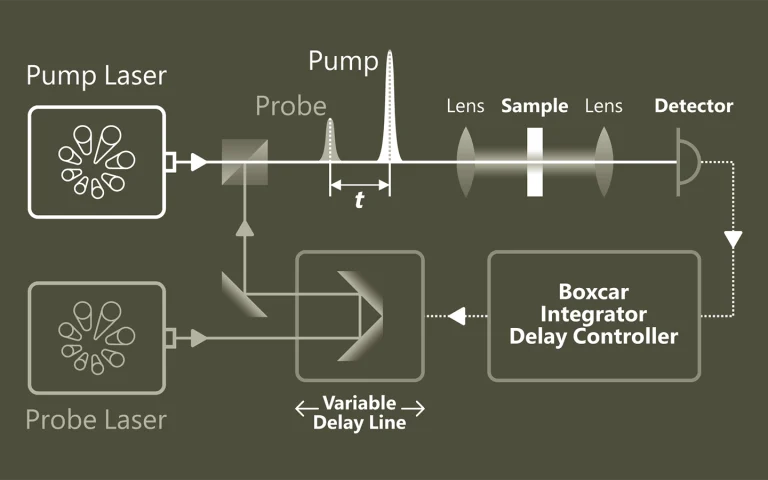
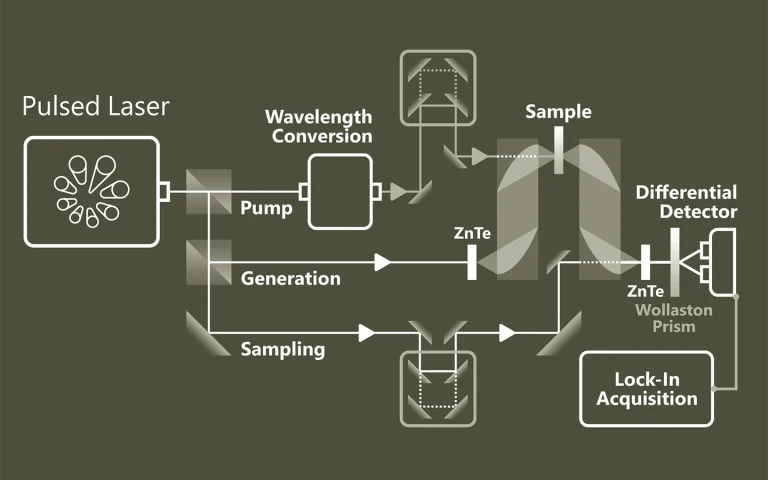
Pump-probe spectroscopy
Ultrafast spectroscopy is based on the use of light pulses that have shorter temporal duration compared to the underlying dynamics of the system. Shorter pulse- better time resolution, longer pulse- better spectral resolution. Two pulses separated by time delay are used. First pulse excites (electronic and/or vibrational levels, ionises, make Coulombic explosions, creates plasma, etc.) sample, second delayed pulse probes- checks what happened at the certain time moment. Dynamics of investigated system is recorded by changing delay between pulses. Various experimental techniques are employed to record time-resolved signals: for example transient absorption, four-wave mixing, sum frequency generation. Pump and probe spectral range can vary from UV or even X-ray to far infrared.
The advantage of pump-probe spectroscopy is direct investigation of dynamics. For example: excitation relaxation, energy transfer, photochemical reactions dynamics and movement of particles, structural changes. Tunability of pump and probe pulses opens two dimensional pump-probe spectroscopy where is possible to obtain temporally resolved energy map of system which shows separated and coupled states.
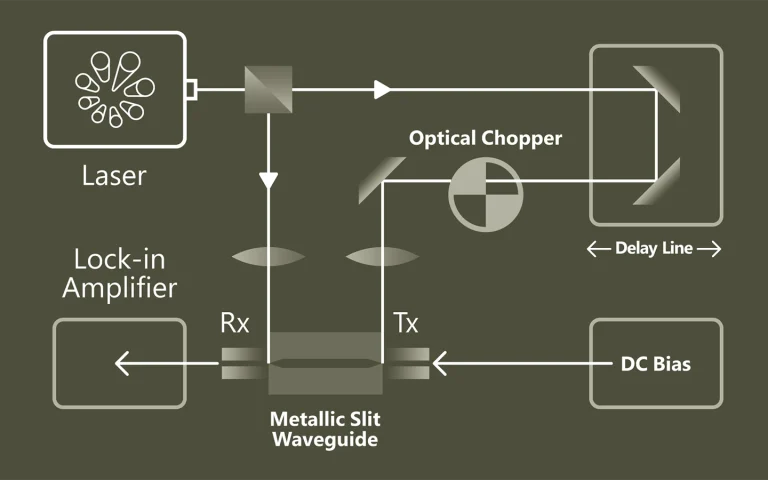
Previous measurements of vibrational relaxation lifetimes for molecules adsorbed at metal surfaces yielded values of 1 – 3 ps; however, only chemisorbed molecules have been studied. We report the first measurements of the vibrational relaxation lifetime of a molecule physisorbed to a metal surface. For CO(υ=1) adsorbed on Au(111) at 35 K the vibrational lifetime of the excited stretching mode is 49±3 ps. The long lifetime seen here is likely to be a general feature of physisorption, which involves weaker electronic coupling between the adsorbate and the solid due to bonding at larger distances.
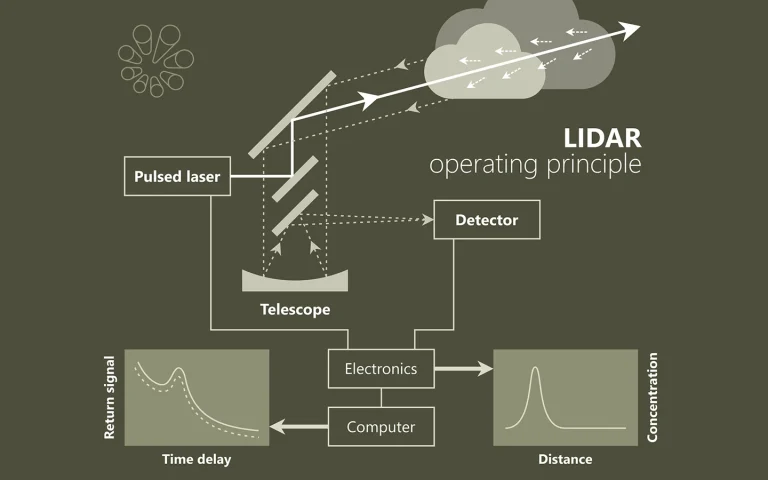
Localized surface plasmon resonance (LSPR)-induced hot-carrier transfer is a key mechanism for achieving artificial photosynthesis using the whole solar spectrum, even including the infrared (IR) region. In contrast to the explosive development of photocatalysts based on the plasmon-induced hot electron transfer, the hole transfer system is still quite immature regardless of its importance, because the mechanism of plasmon-induced hole transfer has remained unclear. Herein, we elucidate LSPR-induced hot hole transfer in CdS/CuS heterostructured nanocrystals (HNCs) using time-resolved IR (TR-IR) spectroscopy. TR-IR spectroscopy enables the direct observation of carrier in a LSPR-excited CdS/CuS HNC. The spectroscopic results provide insight into the novel hole transfer mechanism, named plasmon-induced transit carrier transfer (PITCT), with high quantum yields (19%) and long-lived charge separations (9.2 μs). As an ultrafast charge recombination is a major drawback of all plasmonic energy conversion systems, we anticipate that PITCT will break the limit of conventional plasmon-induced energy conversion.
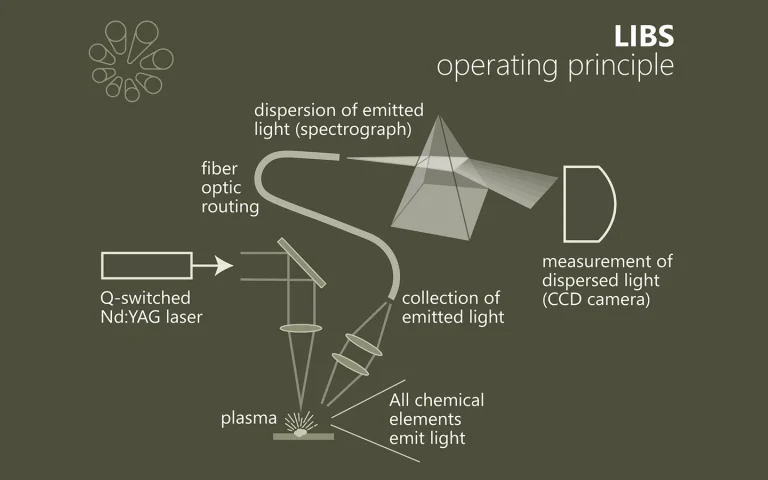
Time-resolved serial femtosecond crystallography using an X-ray free electron laser (XFEL) in conjunction with a photosensitive caged-compound offers a crystallographic method to track enzymatic reactions. Here we demonstrate the application of this method using fungal NO reductase, a heme-containing enzyme, at room temperature. Twenty milliseconds after caged-NO photolysis, we identify a NO-bound form of the enzyme, which is an initial intermediate with a slightly bent Fe-N-O coordination geometry at a resolution of 2.1 Å. The NO geometry is compatible with those analyzed by XFEL-based cryo-crystallography and QM/MM calculations, indicating that we obtain an intact Fe3+-NO coordination structure that is free of X-ray radiation damage. The slightly bent NO geometry is appropriate to prevent immediate NO dissociation and thus accept H− from NADH. The combination of using XFEL and a caged-compound is a powerful tool for determining functional enzyme structures during catalytic reactions at the atomic level.
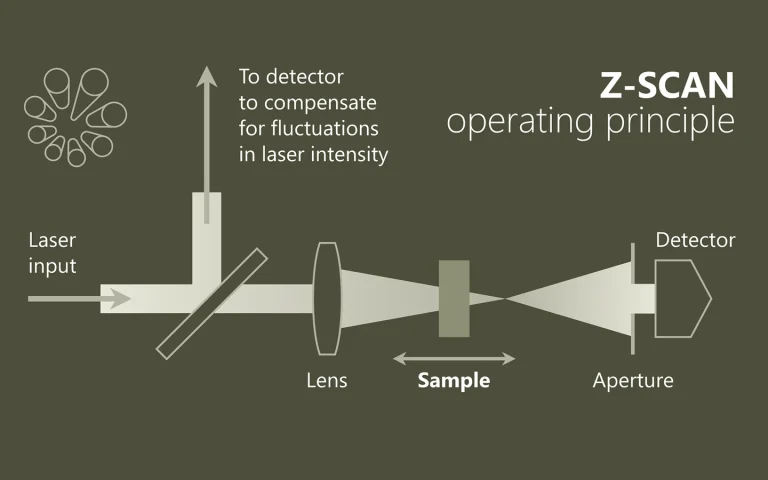
Luminescence upconversion nanocrystals capable of converting two low-energy photons into a single photon at a higher energy are sought-after for a variety of applications, including bioimaging and photovoltaic light harvesting. Currently available systems, based on rare-earth-doped dielectrics, are limited in both tunability and absorption cross-section. Here we present colloidal double quantum dots as an alternative nanocrystalline upconversion system, combining the stability of an inorganic crystalline structure with the spectral tunability afforded by quantum confinement. By tailoring its composition and morphology, we form a semiconducting nanostructure in which excited electrons are delocalized over the entire structure, but a double potential well is formed for holes. Upconversion occurs by excitation of an electron in the lower energy transition, followed by intraband absorption of the hole, allowing it to cross the barrier to a higher energy state. An overall conversion efficiency of 0.1% per double excitation event is achieved.
Benzhydryl radicals and cations are reactive intermediates central to the under-standing of organic reactivity. They can be generated from benzhydryl halides by UV irradi-ation. We performed transient absorption (TA) measurements over the range from femto -seconds to microseconds to unravel the complete reaction scheme. The 290 – 720-nm proberange allows the unambiguous monitoring of all fragments. The appearance of the radical isdelayed to the optical excitation, the onset of the cation signal is found even later. Ab initiocalculations show that this non-rate behavior in the 100 fs range is due to wavepacket motionfrom the Franck–Condon region to two distinct conical intersections. The rise of the opticalsignal with a quasi-exponential time of 300 fs is assigned to the planarization and solvationof the photoproducts. The bond cleavage predominantly generates radical pairs. A subse-quent electron transfer (ET) transforms radical pairs into ion pairs. Due to the broad inter-radical distance distribution and the distance dependence, the ET is strongly non-exponen-tial. Part of the ion pairs recombine geminately. The ET and the recombination are terminatedby the depletion of close pairs and diffusional separation. The remaining free radicals and cations undergo further reactions in the nanosecond to microsecond regime.