NT230 series High Energy Broadly Tunable DPSS Lasers
NT230 series tunable wavelength laser offers hands free, no-gap tuning from 192 to 2600 nm. from the one box. With its 100 Hz repetition rate, the NT230 series laser establishes itself as a versatile tool for many laboratory applications, as laser induced fluorescence, flash photolysis, photobiology, metrology, remote sensing.
- Customers recognized reliability
- Two years warranty
- Integrates DPSS pump laser and OPO into a single housing
- Hands-free no-gap wavelength tuning from 192 to 2600 nm*
- Up to 15 mJ pulse energy from OPO
- Up to 100 Hz pulse repetition rate
- Up to 2 mJ output pulse energy in UV
- Less than 5 cm⁻¹ linewidth
- 2 – 5 ns pulse duration
- Electromechanical output shutters
- Transportation handles
- 355 nm & 1064 nm laser outputs
- 532 nm output (optional)
- Remote control via key pad or PC
* Automatic wavelength scan is programmable
Benefits
- Hands-free wavelength tuning – no need for physical intervention
- The system is widely tunable; 192 – 2600 nm and delivers high pulse energy (up to 15 mJ) which allows investigation of an extensive range of materials
- High repetition rate (up to 100 Hz) and output power enable fast data collection and intensive excitation of materials
- Narrow linewidth (down to 3 cm‑1) and superior tuning resolution (1 – 2 cm‑1) allow recording of high quality spectra
- High integration level saves valuable space in the laboratory
- Diode pumping reduces maintenance frequency
- Automatic electromechanical output shutters ensure high level of safety
- User friendly extendable handles ease transportation and repositioning of laser
- In-house design and manufacturing of complete systems, including pump lasers, guarantees on-time warranty and post warranty services and spares supply
- Variety of control interfaces: USB, RS232, LAN and WLAN ensures easy control and integration with other equipment
- Attenuator and fiber coupling options facilitate incorporation of NT230 systems into various experimental environments
Applications
- Laser-induced fluorescence
- Flash photolysis
- Photobiology
- Remote sensing
- Metrology
- Non-linear spectroscopy
- Photo acoustic imaging
NT230 series lasers deliver high up to 10 mJ energy pulses at 100 Hz pulse repetition rate, tunable over a broad spectral range. Integrated into a single compact housing, the diode pumped Q-switched Nd:YAG laser and optical parameteric oscillator (OPO) offers hands free, no-gap tuning from 192 to 2600 nm. With its 100 Hz repetition rate, the NT230 series laser establishes itself as a versatile tool for many laboratory applications, as laser induced fluorescence, flash photolysis, photobiology, metrology, remote sensing, etc.
Due to the innovative diode pumped design, NT230 series lasers feature maintenance-free laser operation for an extended period of time and improved stability (compared with flash-lamp pumped counterparts).
NT230 series systems can be controlled from a remote control pad or/and a computer. The control pad allows easy control of all parameters and features on a backlit system display that is easy to read even with laser safety eyewear.
| Model | NT230-50 | NT230-100 |
|---|---|---|
| OPO specifications 1) | ||
| Wavelength range | ||
| Signal | 405 – 710 nm | 405 – 710 nm |
| Idler | 710 – 2600 nm | 710 – 2600 nm |
| SH and SF | 210 – 405 nm 2) | 210 – 405 nm 2) |
| DUV | 192 – 210 nm | 192 – 210 nm |
| Pulse energy 3) | ||
| OPO | 15 mJ | 10 mJ |
| SH and SF 4) | 1.8 mJ | 1.3 mJ |
| DUV | 0.4 mJ | 0.27 mJ |
| Pulse repetition rate | 50 Hz | 100 Hz |
| Pulse duration 5) | 2 – 5 ns | 2 – 5 ns |
| Linewidth 6) | < 5 cm‑1 | < 5 cm‑1 |
| Minimal tuning step 7) | ||
| Signal | 1 cm‑1 | 1 cm‑1 |
| Idler | 1 cm‑1 | 1 cm‑1 |
| SH / SF / DUV | 2 cm‑1 | 2 cm‑1 |
| Polarization | ||
| Signal | horizontal | horizontal |
| Idler | vertical | vertical |
| SH / SF | horizontal | horizontal |
| DUV | vertical | vertical |
| OPO beam divergence 8) | < 2 mrad | < 2 mrad |
| Typical beam diameter 9) | 5 mm | 5 mm |
| Pump laser | ||
| Pump wavelength 10) | 355 nm | 355 nm |
| Typical pump pulse energy 11) | 50 mJ | 35 mJ |
| Pulse duration | 2 – 5 ns | 2 – 5 ns |
| Physical characteristics | ||
| Unit size (W × L × H) 12) | 451 × 705 × 172 mm | 451 × 705 × 172 mm |
| Power supply size (W × L × H) | 449 × 376 × 140 mm | 449 × 376 × 140 mm |
| External chiller | inquire | inquire |
| Umbilical length | 2.5 m | 2.5 m |
| Operating requirements | ||
| Cooling | external chiller | external chiller |
| Room temperature | 18 – 30 °C | 18 – 30 °C |
| Relative humidity | 20 – 80 % (non-condensing) | 20 – 80 % (non-condensing) |
| Power requirements | 100 – 240 V AC, single phase, 50/60 Hz | 100 – 240 V AC, single phase, 50/60 Hz |
| Power consumption | < 1.8 kW | < 1.8 kW |
| Cleanliness of the room | not worse than ISO Class 9 | not worse than ISO Class 9 |
| Model | NT230-50 | NT230-100 |
|---|
- Due to continuous improvement, all specifications are subject to change. Parameters marked typical are illustrative. They are indications of typical performance and will vary with each unit we manufacture. Unless stated otherwise, all specifications are measured at 450 nm and for basic system without options.
- Separate –SH and –SF options are available.
- See tuning curves for typical outputs at other wavelengths.
- Measured at 260 nm wavelength.
- FWHM measured with photodiode featuring 1 ns rise time and 300 MHz bandwidth oscilloscope.
- Linewidth is <8 cm‑1 for 210 – 405 nm range.
- When wavelength is controlled from PC. When wavelength is controlled from keypad, tuning resolution is 0.1 nm for signal, 1 nm for idler and 0.05 nm for SH, SF and DUV.
- Full angle measured at the FWHM level at 450 nm.
- Beam diameter is measured at 450 nm at the 1/e2 level and can vary depending on the pump pulse energy.
- Separate output port for the fundamental and 3rd harmonic beam is standard. Output ports for other harmonic are optional.
- The pump laser pulse energy will be optimized for best OPO performance and can vary with each unit we manufacture.
- Length from 705 to 1016 mm depending on configuration.
Note: Laser must be connected to the mains electricity all the time. If there will be no mains electricity for longer that 1 hour then laser (system) needs warm up for a few hours before switching on.
| Option | Features |
|---|---|
| -SH | Tuning range extension in UV range (210 – 405 nm) by second harmonic generation |
| -SF | Tuning range extension in 300 – 405 nm range by sum-frequency generation |
| -SH/SF | Tuning range extension in 210 – 405 nm range by combining second harmonic and sum-frequency generator outputs for maximum possible pulse energy |
| -DUV | Deep UV option for 192 – 210 nm range output |
| -2H | 532 nm output |
| -FC | Fiber coupled output in 300 – 2000 nm range |
| -ATTN | Attenuator |
| -SCU | Spectral filtering accessory for improved spectral purity of pulses |
| -FWS | Fast wavelength scanning option |
| Option | Features |
|---|
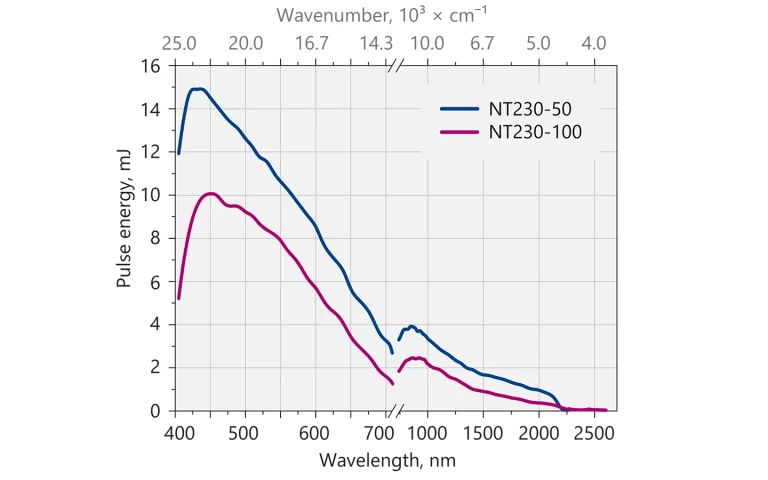
Typical output pulse energy of NT230 laser.
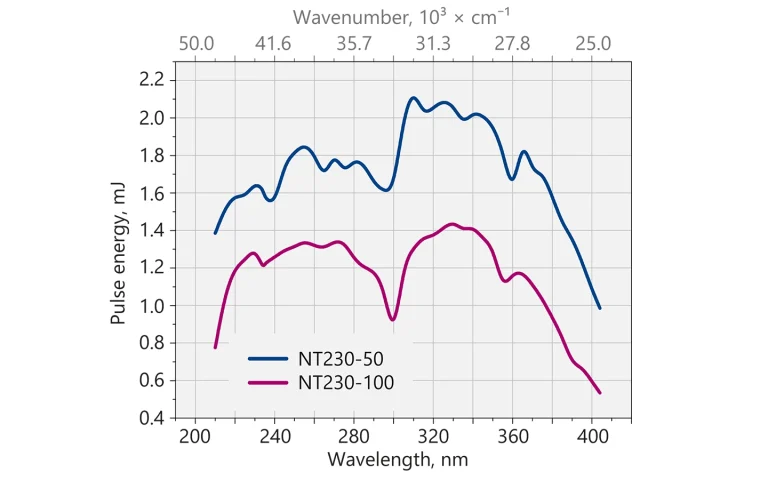
Typical output pulse energy of NT230 laser with SH/SF extension.
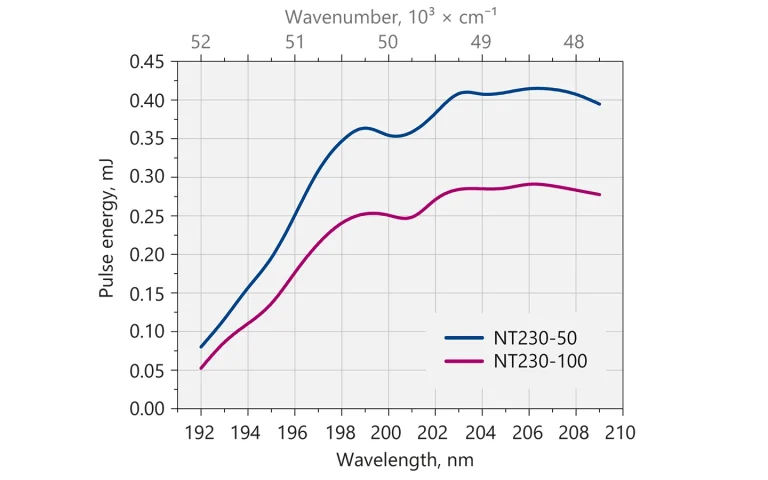
Typical output pulse energy of NT230 laser with DUV extension.
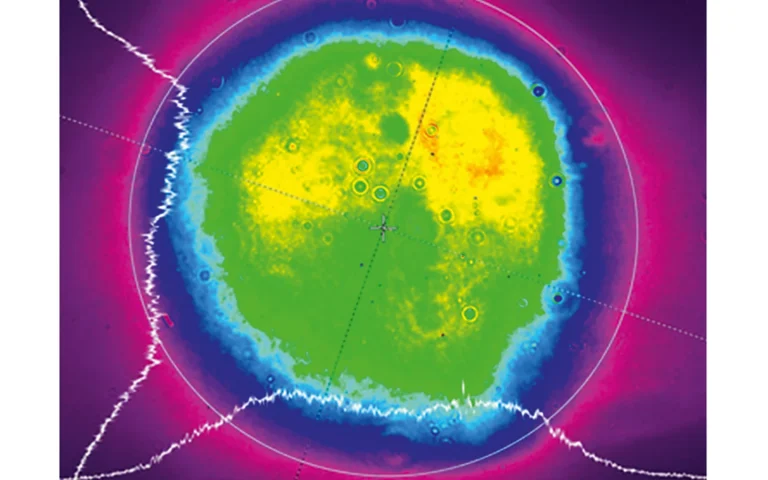
Typical beam profile of NT230 series lasers.
Near field at 450 nm.
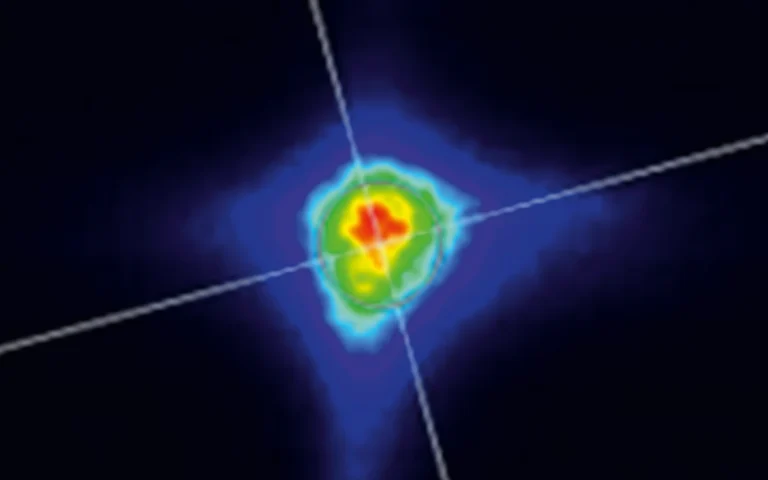
Typical beam profile of NT230 series lasers.
Far field at 450 nm.
Hydrated singly charged magnesium ions [Mg(H2O)n]+ are thought to consist of an Mg2+ ion and a hydrated electron for n > 15. This idea is based on mass spectra, which exhibit a transition from [MgOH(H2O)n−1]+ to [Mg(H2O)n]+ around n = 15 – 22, black-body infrared radiative dissociation, and quantum chemical calculations. Here, we present photodissociation spectra of size-selected [Mg(H2O)n]+ in the range of n = 20 – 70 measured for photon energies of 1.0 – 5.0 eV. The spectra exhibit a broad absorption from 1.4 to 3.2 eV, with two local maxima around 1.7 – 1.8 eV and 2.1 – 2.5 eV, depending on cluster size. The spectra shift slowly from n = 20 to n = 50, but no significant change is observed for n = 50 – 70. Quantum chemical modeling of the spectra yields several candidates for the observed absorptions, including five- and six-fold coordinated Mg2+ with a hydrated electron in its immediate vicinity, as well as a solvent-separated Mg2+/e− pair. The photochemical behavior resembles that of the hydrated electron, with barrierless interconversion into the ground state following the excitation.
Here we report gas-phase action and luminescence spectra of cationic dyes derived from oxazine: cresyl violet (CV+), oxazine 170 (Ox-170+), nile blue (NB+), darrow red (DR+), oxazine 1 (Ox-1+), oxazine 4 (Ox-4+), and brilliant cresyl blue (BCB+). The first four have a benzofused structure, which results in asymmetric charge distributions along the long axis. The positive charge is also asymmetrically distributed in BCB+ while Ox-1+ and Ox-4+ are symmetric. As the ions are isolated in vacuo, there are no interactions with solvent molecules or counter ions, and the effect of chemical modifications is therefore more easily revealed than from solution-phase experiments. The transition energy decreases in the order: DR+ > CV+ > Ox-4+ > Ox-170+ > BCB+ > Ox-1+ > NB+, and the fluorescence from BCB+ is less than from the others. We discuss the results based on electron delocalisation, degree of charge-transfer character, rigidity of the chromophore structure, and substituents.
Hydrated singly charged magnesium ions Mg+(H2O)n, n ≤ 5, in the gas phase are ideal model systems to study photochemical hydrogen evolution since atomic hydrogen is formed over a wide range of wavelengths, with a strong cluster size dependence. Mass selected clusters are stored in the cell of an Fourier transform ion cyclotron resonance mass spectrometer at a temperature of 130 K for several seconds, which allows thermal equilibration via blackbody radiation. Tunable laser light is used for photodissociation. Strong transitions to D1 – 3 states (correlating with the 3s-3px,y,z transitions of Mg+) are observed for all cluster sizes, as well as a second absorption band at 4 – 5 eV for n = 3-5. Due to the lifted degeneracy of the 3px,y,z energy levels of Mg+, the absorptions are broad and red shifted with increasing coordination number of the Mg+ center, from 4.5 eV for n = 1 to 1.8 eV for n = 5. In all cases, H atom formation is the dominant photochemical reaction channel. Quantum chemical calculations using the full range of methods for excited state calculations reproduce the experimental spectra and explain all observed features. In particular, they show that H atom formation occurs in excited states, where the potential energy surface becomes repulsive along the O⋯H coordinate at relatively small distances. The loss of H2O, although thermochemically favorable, is a minor channel because, at least for the clusters n = 1-3, the conical intersection through which the system could relax to the electronic ground state is too high in energy. In some absorption bands, sequential absorption of multiple photons is required for photodissociation. For n = 1, these multiphoton spectra can be modeled on the basis of quantum chemical calculations.
Marine aerosols consist of a variety of compounds and play an important role in many atmospheric processes. In the present study, sodium iodide clusters with their simple isotope pattern serve as model systems for laboratory studies to investigate the role of iodide in the photochemical processing of sea-salt aerosols. Salt clusters doped with camphor, formate and pyruvate are studied in a Fourier transform ion cyclotron resonance mass spectrometer (FT-ICR MS) coupled to a tunable laser system in both UV and IR range. The analysis is supported by ab initio calculations of absorption spectra and energetics of dissociative channels. We provide quantitative analysis of IRMPD measurements by reconstructing one-photon spectra and comparing them with the calculated ones. While neutral camphor is adsorbed on the cluster surface, the formate and pyruvate ions replace an iodide ion. The photodissociation spectra revealed several wavelength-specific fragmentation pathways, including the carbon dioxide radical anion formed by photolysis of pyruvate. Camphor and pyruvate doped clusters absorb in the spectral region above 290 nm, which is relevant for tropospheric photochemistry, leading to internal conversion followed by intramolecular vibrational redistribution, which leads to decomposition of the cluster. Potential photodissociation products of pyruvate in the actinic region may be formed with a cross section of <2×10−20 cm2, determined by the experimental noise level.
Time-resolved serial femtosecond crystallography using an X-ray free electron laser (XFEL) in conjunction with a photosensitive caged-compound offers a crystallographic method to track enzymatic reactions. Here we demonstrate the application of this method using fungal NO reductase, a heme-containing enzyme, at room temperature. Twenty milliseconds after caged-NO photolysis, we identify a NO-bound form of the enzyme, which is an initial intermediate with a slightly bent Fe-N-O coordination geometry at a resolution of 2.1 Å. The NO geometry is compatible with those analyzed by XFEL-based cryo-crystallography and QM/MM calculations, indicating that we obtain an intact Fe3+-NO coordination structure that is free of X-ray radiation damage. The slightly bent NO geometry is appropriate to prevent immediate NO dissociation and thus accept H− from NADH. The combination of using XFEL and a caged-compound is a powerful tool for determining functional enzyme structures during catalytic reactions at the atomic level.
We report a target-enclosing, hybrid tomograph with a total of 768 elements based on capacitive micromachined ultrasound transducer technology and providing fast, high-resolution 2-D/3-D photoacoustic and ultrasound tomography tailored to finger imaging.A freely programmable ultrasound beamforming platform sampling data at 80 MHz was developed to realize plane wave transmission under multiple angles. A multiplexing unit enables the connection and control of a large number of elements. Fast image reconstruction is provided by GPU processing. The tomograph is composed of four independent and fully automated movable arc-shaped transducers, allowing imaging of all three finger joints. The system benefits from photoacoustics, yielding high optical contrast and enabling visualization of finger vascularization, and ultrasound provides morphologic information on joints and surrounding tissue. A diode-pumped, Q-switched Nd:YAG laser and an optical parametric oscillator are used to broaden the spectrum of emitted wavelengths to provide multispectral imaging. Custom-made optical fiber bundles enable illumination of the region of interest in the plane of acoustic detection. Precision in positioning of the probe in motion is ensured by use of a motor-driven guide slide. The current position of the probe is encoded by the stage and used to relate ultrasound and photoacoustic signals to the corresponding region of interest of the suspicious finger joint. The system is characterized in phantoms and a healthy human finger in vivo. The results obtained promise to provide new opportunities in finger diagnostics and establish photoacoustic/ultrasoundtomography in medical routine.
Protonated leucine enkephalin (YGGFL) was studied by ultraviolet photodissociation (UVPD) from 225 to 300 nm utilizing an optical parametric oscillator tunable wavelength laser system (OPO). Fragments were identified by absolute mass measurement in a 9.4 T Fourier transform ion cyclotron resonance mass spectrometer (FT-ICR MS). Bond cleavage was preferred in the vicinity of the two aromatic residues, resulting in high ion abundances for a4, a1, b3, y2 and y1 fragments. a, b and y ions dominated the mass spectrum, and full sequence coverage was achieved for those types. Photodissociation was most effective at the short wavelength end of the studied range, which is assigned to the onset of the La π–π* transition of the tyrosine chromophore, but worked well also at the Lb π–π* chromophore absorption maxima in the 35 000 – 39 000 cm−1 region. Several side-chain and internal fragments were observed. H atom loss is observed only above 41 000 cm−1, consistent with the requirement of a curve crossing to a repulsive 1πσ* state. It is suggested that the photochemically generated mobile H atom plays a role in further backbone cleavages, similar to the mechanism for electron capture dissociation. The b4 fragment is most intense at the Lb chromophore absorptions, undergoing additional fragmentation at higher photon energies. The high resolution of the FT-ICR MS revealed that out of all x and z-type fragments only x3 and x4 were formed, with low intensity. Other previously reported x- and z-fragments were re-assigned to internal fragments, based on exact mass measurement.
A relatively simple setup for collection and detection of light emitted from isolated photo-excited molecular ions has been constructed. It benefits from a high collection efficiency of photons, which is accomplished by using a cylindrical ion trap where one end-cap electrode is a mesh grid combined with an aspheric condenser lens. The geometry permits nearly 10% of the emitted light to be collected and, after transmission losses, approximately 5% to be delivered to the entrance of a grating spectrometer equipped with a detector array. The high collection efficiency enables the use of pulsed tunable lasers with low repetition rates (e.g., 20 Hz) instead of continuous wave (cw) lasers or very high repetition rate (e.g., MHz) lasers that are typically used as light sources for gas-phase fluorescence experiments on molecular ions. A hole has been drilled in the cylinder electrode so that a light pulse can interact with the ion cloud in the center of the trap. Simulations indicate that these modifications to the trap do not significantly affect the storage capability and the overall shape of the ion cloud. The overlap between the ion cloud and the laser light is basically 100%, and experimentally >50% of negatively charged chromophore ions are routinely photodepleted. The performance of the setup is illustrated based on fluorescence spectra of several laser dyes, and the quality of these spectra is comparable to those reported by other groups. Finally, by replacing the optical system with a channeltron detector, we demonstrate that the setup can also be used for gas-phase action spectroscopy where either depletion or fragmentation is monitored to provide an indirect measurement on the absorption spectrum of the ion.
Benzhydryl radicals and cations are reactive intermediates central to the under-standing of organic reactivity. They can be generated from benzhydryl halides by UV irradi-ation. We performed transient absorption (TA) measurements over the range from femto -seconds to microseconds to unravel the complete reaction scheme. The 290 – 720-nm proberange allows the unambiguous monitoring of all fragments. The appearance of the radical isdelayed to the optical excitation, the onset of the cation signal is found even later. Ab initiocalculations show that this non-rate behavior in the 100 fs range is due to wavepacket motionfrom the Franck–Condon region to two distinct conical intersections. The rise of the opticalsignal with a quasi-exponential time of 300 fs is assigned to the planarization and solvationof the photoproducts. The bond cleavage predominantly generates radical pairs. A subse-quent electron transfer (ET) transforms radical pairs into ion pairs. Due to the broad inter-radical distance distribution and the distance dependence, the ET is strongly non-exponen-tial. Part of the ion pairs recombine geminately. The ET and the recombination are terminatedby the depletion of close pairs and diffusional separation. The remaining free radicals and cations undergo further reactions in the nanosecond to microsecond regime.